 What is the Difference Between Hydrogen and Helium Emission Spectra? However, since residing in a high energy level is unstable, these electrons tend to come back to the ground level (energy level at which they existed previously) emitting a photon as electromagnetic radiation that has an energy equal to the energy difference between these higher and lower energy levels. After that, the electron in the hydrogen atom gets excited to a higher energy level. There, when we pass a beam of white light through a sample of hydrogen gas, then the atoms absorb energy. The hydrogen emission spectrum is a spectrum produced by the emission of light by hydrogen atoms in excited states. Summary What is Hydrogen Emission Spectra? 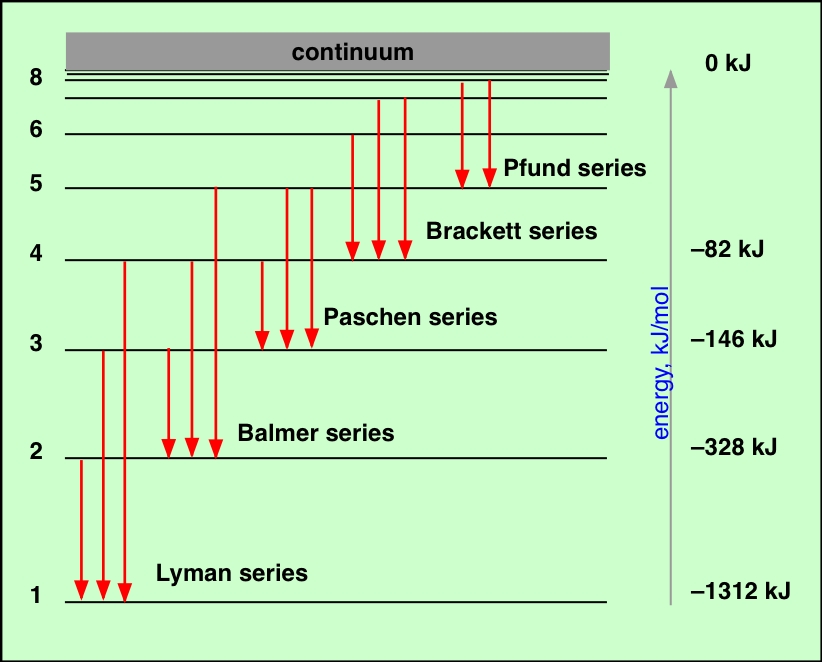
Side by Side Comparison – Hydrogen vs Helium Emission Spectra in Tabular Formĥ. The emission spectrum of a chemical element or compound is the series of lines that represent the wavelengths of electromagnetic radiation emitted by that chemical element while the transition of an electron from a high energy level to a low energy level. spectra) has more lines than that of the hydrogen emission spectrum (plu. Bohr's model and postulates do not explain this phenomenon.The key difference between hydrogen and helium emission spectra is that the helium emission spectrum (plu. Spectral lines undergo splitting when a sample of gas is affected by an electric field (Stark effect) and magnetic field (Zeeman effect).These are referred to as hyperfine spectral lines. Bohr's model does not explain the presence of hyperfine lines. 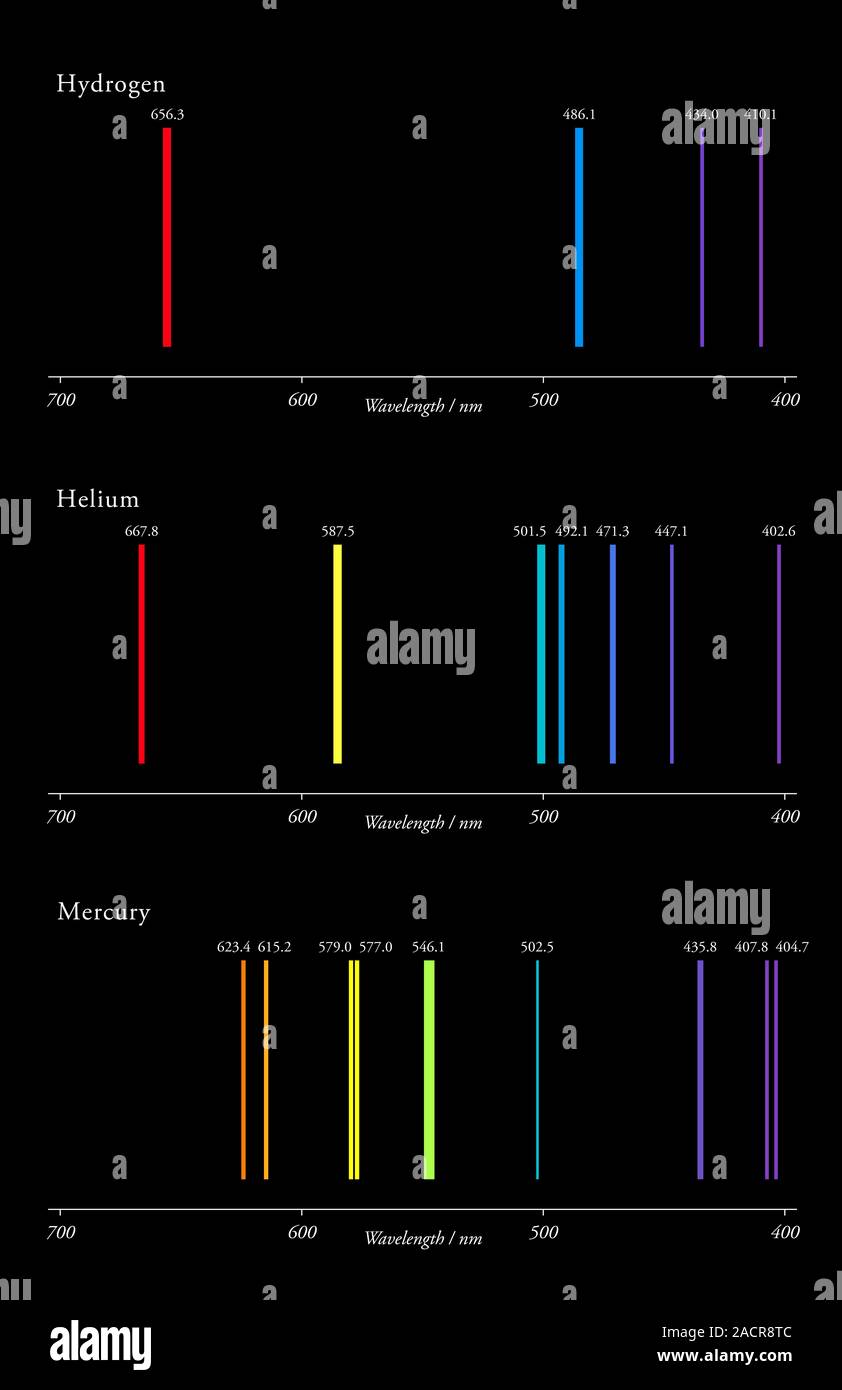
Each spectral line, when examined at higher magnification, actually consists of a number of smaller fine lines. 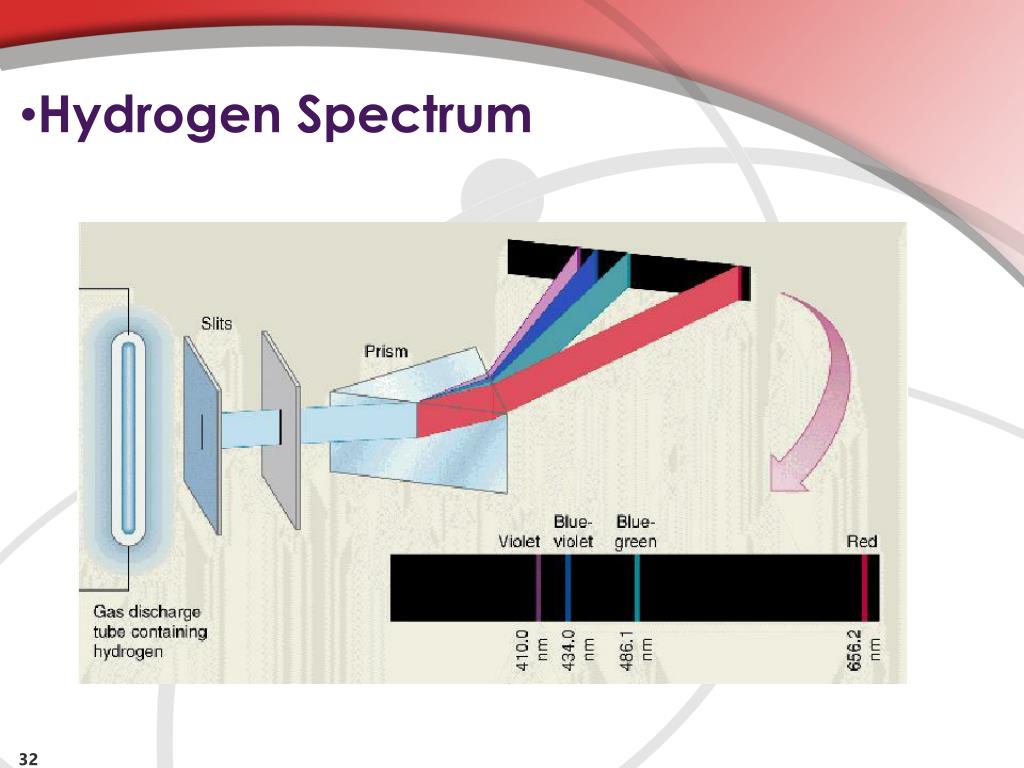
The accuracy decreases as the effective nuclear charge of an atom or ion increases (due to greater number of protons). Predictions made by Bohr’s model is only accurate for the hydrogen atom.Bohr's model cannot predict the relative intensity (brightness) of the spectral lines.In other words, his model does not explain why electrons can remain in their orbits without spiralling into the nucleus due to electrostatic attraction. Bohr does not provide an explanation to 'stationary states' of electrons. 
Circular motion of electrons is from classical physics while the quantisation of its momentum and energy of orbits is from quantum physics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
